SmartPeg Reference Guide
Your complete reference for choosing the correct SmartPeg for your implant system.
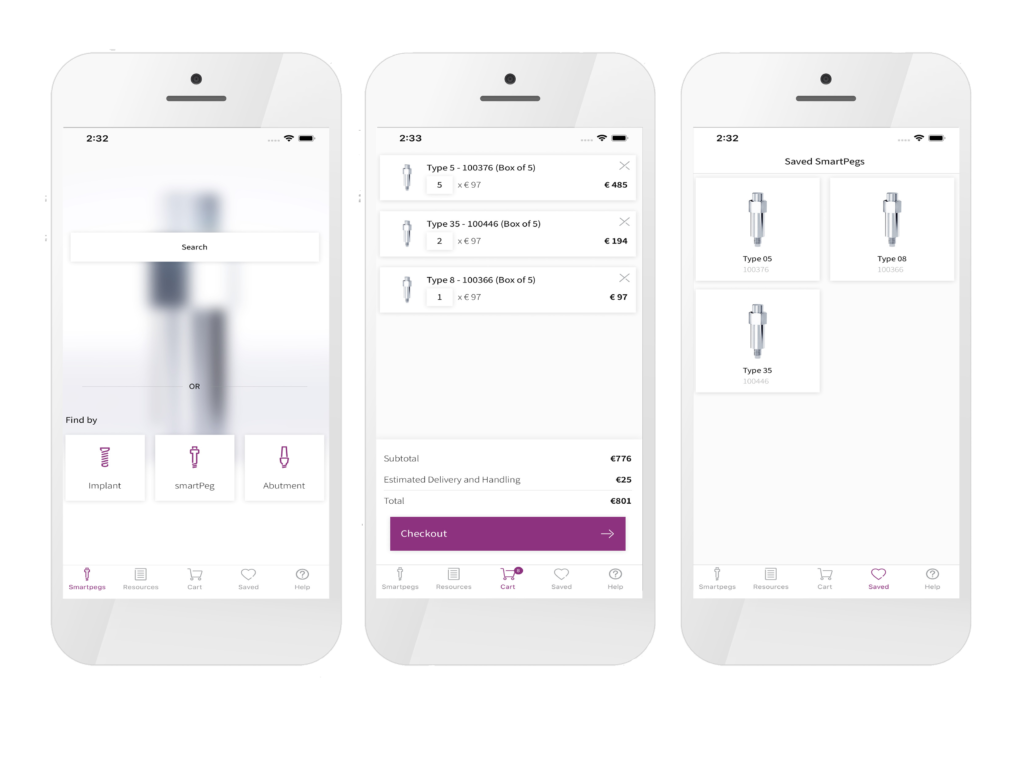
The SmartPeg Reference Guide helps you identify the specific SmartPeg type required – now showing both single‑use and reusable SmartPegs side by side, to help you select the option that best fits your clinical workflow.
Find your distributor
You will find a complete list of all our local distributors
Understanding what SmartPegs measure
ISQ (Implant Stability Quotient) is a scale from 1 to 99 used to assess the stability of dental implants. The ISQ value is derived from a measurement of implant micromobility: the lower the micromobility, the higher the resulting stability. Because it is more clinically meaningful to interpret stability rather than mobility, this micromobility measurement is converted into an ISQ value – providing a clear indication of implant stability and supporting knowledge‑based treatment decisions. Supported by more than 1,500 scientific references, ISQ values are commonly interpreted as:
- High stability: ISQ >70
- Medium stability: ISQ 60–69
- Low stability: ISQ <60
SmartPegs are the precision‑engineered components required to obtain these values. When attached to the implant or abutment, they enable the Osstell instrument to measure resonance frequency and convert it into an objective, repeatable ISQ value.
Why SmartPegs are single use or reusable – and why it matters
Reusable SmartPeg reprocessing quickguide
Is the ISQ value affected by the torque of the SmartPeg?
The SmartPeg should be tightened with 4-6 Ncm, which means “finger tight”, using the SmartPeg Mount. It is enough with firm contact between the SmartPeg and the implant. The difference in ISQ value will then stay within 2 ISQ units. (If the SmartPeg is tightened too loosely, the ISQ-value will be lower.) If the SmartPeg is over-tightened, there is a risk that the SmartPeg threads will be damaged or that the SmartPeg will fracture.
Did you know that implant stability can be measured at abutment level?
This requires a SmartPeg designed for that purpose, and the results are not directly comparable to those measured at implant level. To establish a reliable baseline, it’s recommended to measure stability both before and after the abutment is installed – during the same treatment visit – so that future follow-up measurements can be accurately interpreted.
Measuring implant stability at the abutment level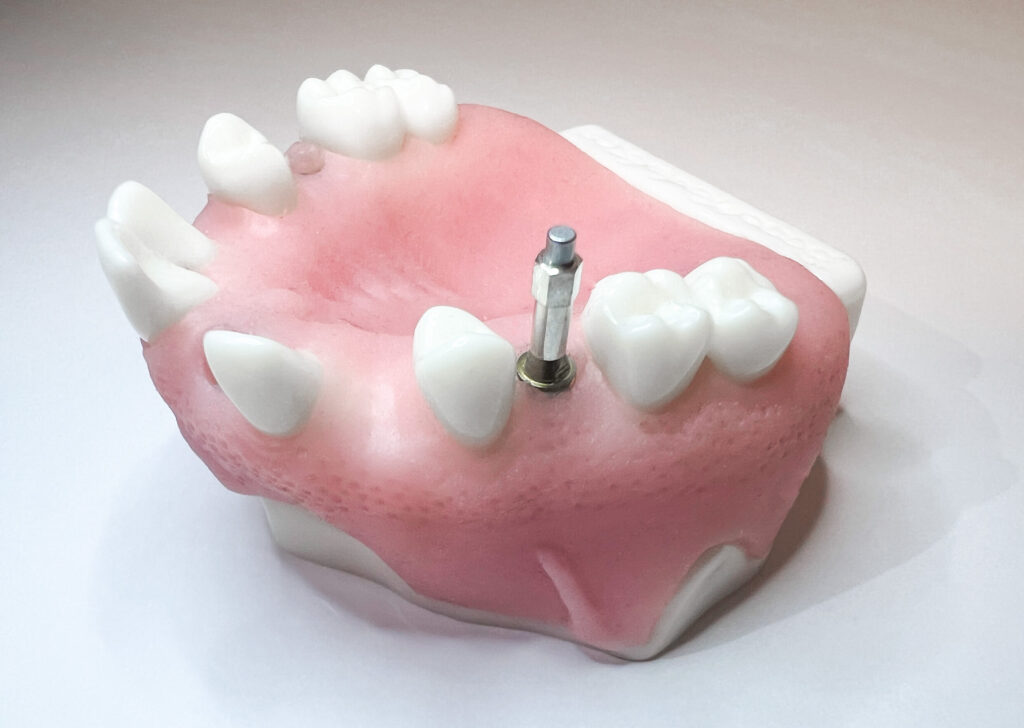
If you get no reading or an unexpected ISQ value
If you get no ISQ reading:
- Check that the correct SmartPeg Type is used.
- Ensure it is tightened finger‑tight (4–6 Ncm) and installed with the SmartPeg Mount.
- Verify that both the implant or abutment surface and the SmartPeg are clean, and ensure the SmartPeg is free from soft‑tissue contact.
- Use only SmartPegs in good condition – new (single‑use) or undamaged and within 20 cycles (reusable).
- Ensure the SmartPeg is an original Osstell SmartPeg.
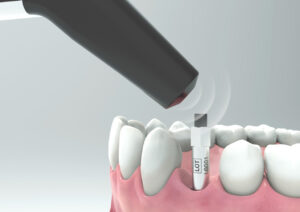
- Ensure the instrument tip is held 2–4 mm above the magnet and angled at approximately 45°.
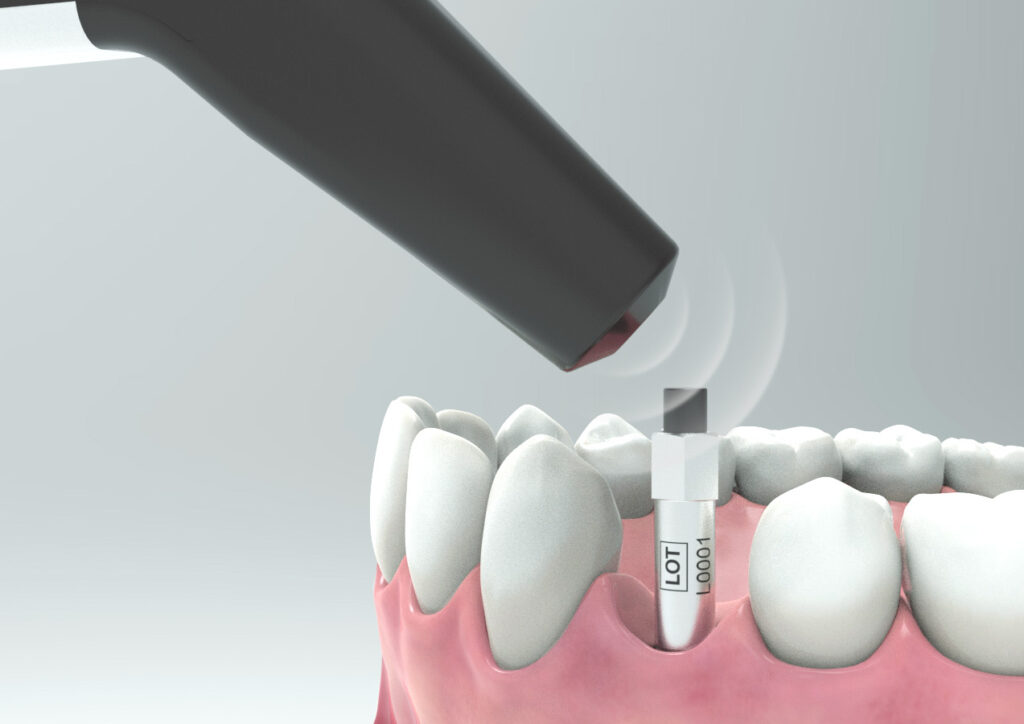
Can I reuse the SmartPegs?
SmartPegs are now available in two versions: single‑use and reusable. Both versions are made from aluminium, ensuring they are gentle on the implant’s internal threads.
The single‑use SmartPegs are designed for one procedure only and cannot be reprocessed. The magnet cannot withstand autoclave temperatures and will become affected or discoloured, and repeated tightening may wear the aluminium threads, potentially leading to inaccurate ISQ values.
The reusable SmartPegs are specifically engineered to tolerate up to 20 validated cleaning and sterilisation procedures. They feature a surface coating that protects both the magnet and the aluminium threads during repeated use, ensuring consistent and reliable ISQ measurements over multiple cycles.
The two versions are easy to distinguish visually:
The single‑use SmartPeg is aluminium‑coloured throughout.
The reusable SmartPeg has a black magnet and black laser‑marked Type and LOT.
Always follow the handling and sterilisation instructions for the version you are using to ensure safe and accurate ISQ measurements.


Frequently asked questions about SmartPegs
Still have questions?
We’re here to help! Simply fill out the form and we’ll contact you as soon as possible.