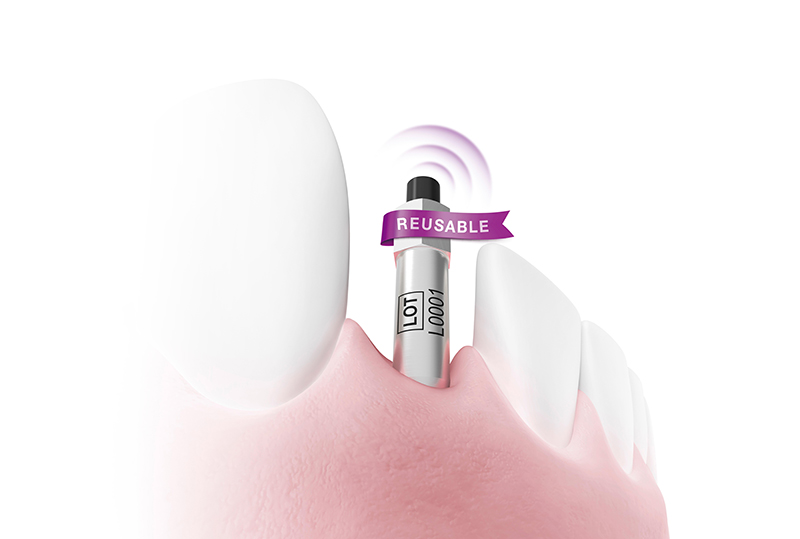
Osstell, a W&H company and the global leader in implant stability measurement, announces the launch of the new reusable SmartPeg, extending its established SmartPeg portfolio. The new version complements the sterile single‑use SmartPeg and offers clinicians greater flexibility in
measuring implant stability – while maintaining the proven accuracy of the ISQ method.
SmartPegs are part of the original Osstell ISQ concept, using the proven RFA method for measuring implant stability. The reusable SmartPeg serves as a complementary extension to the system, using the same validated aluminium design cited in more than 1,500 scientific studies and delivering identical ISQ accuracy. Designed for up to 20 reprocessing cycles, the reusable SmartPeg offers a cost‑efficient alternative. It features a protective surface coating for durable performance, a laser marked type and LOT number for clear identification, and a black magnet for easy measurement orientation and clear distinction from the sterile single-use version.
“With the reusable SmartPeg, we wanted to give dentists more options without changing the way they work or the accuracy they rely on,” says Malin Gustavsson, CEO at Osstell. “It simply adds flexibility, and value while preserving the scientific integrity for which Osstell is known.”
Two SmartPeg options. One validated ISQ method.
With both reusable and single‑use SmartPegs available, Osstell now provides a unified, future‑ready portfolio that adapts to various workflows. All Osstell devices, including Osstell Beacon, Osstell Classic, and earlier systems such as IDx, are compatible with both SmartPeg versions. Dentists can choose the solution that best fits their practice, knowing that each delivers reliable, validated ISQ measurements.
About Osstell
Osstell is the global leader in implant stability measurement. Since the company’s founding in 1999, over thousands of scientific studies have been published confirming the clinical value of Osstell’s proprietary Resonance Frequency Analysis (RFA) technology and the Osstell ISQ scale (Implant Stability Quotient). In 2018, Osstell became a part of the W&H Group, a global leader in the development and manufacture of medical technology products with over 1,300 employees. Osstell is headquartered in Gothenburg, Sweden, the birthplace of dental implants. Our product solution is available in more than 70 countries, through our own presence and a vast network of distributors worldwide.
We empower dental professionals by providing objective implant stability measurements to provide each patient with optimal time to teeth and improve patient confidence.
